What to Expect During a Medical Integration Middleware Implementation
In today’s healthcare world, having clinical systems, enterprise software, and medical devices talk to each other isn't just a "nice to have" anymore—it’s the heartbeat of the operation.
The reality, though, is that many organizations are still fighting with "digital silos." Between clunky legacy platforms, mismatched protocols, and way too many manual workarounds, things get slowed down, risks go up, and trying to modernize feels like pulling teeth.
That’s where medical integration middleware comes in. Think of it as the central nervous system for your tech stack. next.
What should we realistically expect during an implementation?
Understanding the full lifecycle of a middleware implementation helps enterprise leaders plan effectively, allocate resources appropriately, and reduce risk during modernization initiatives.
Middleware Implementation as a Strategic Transformation
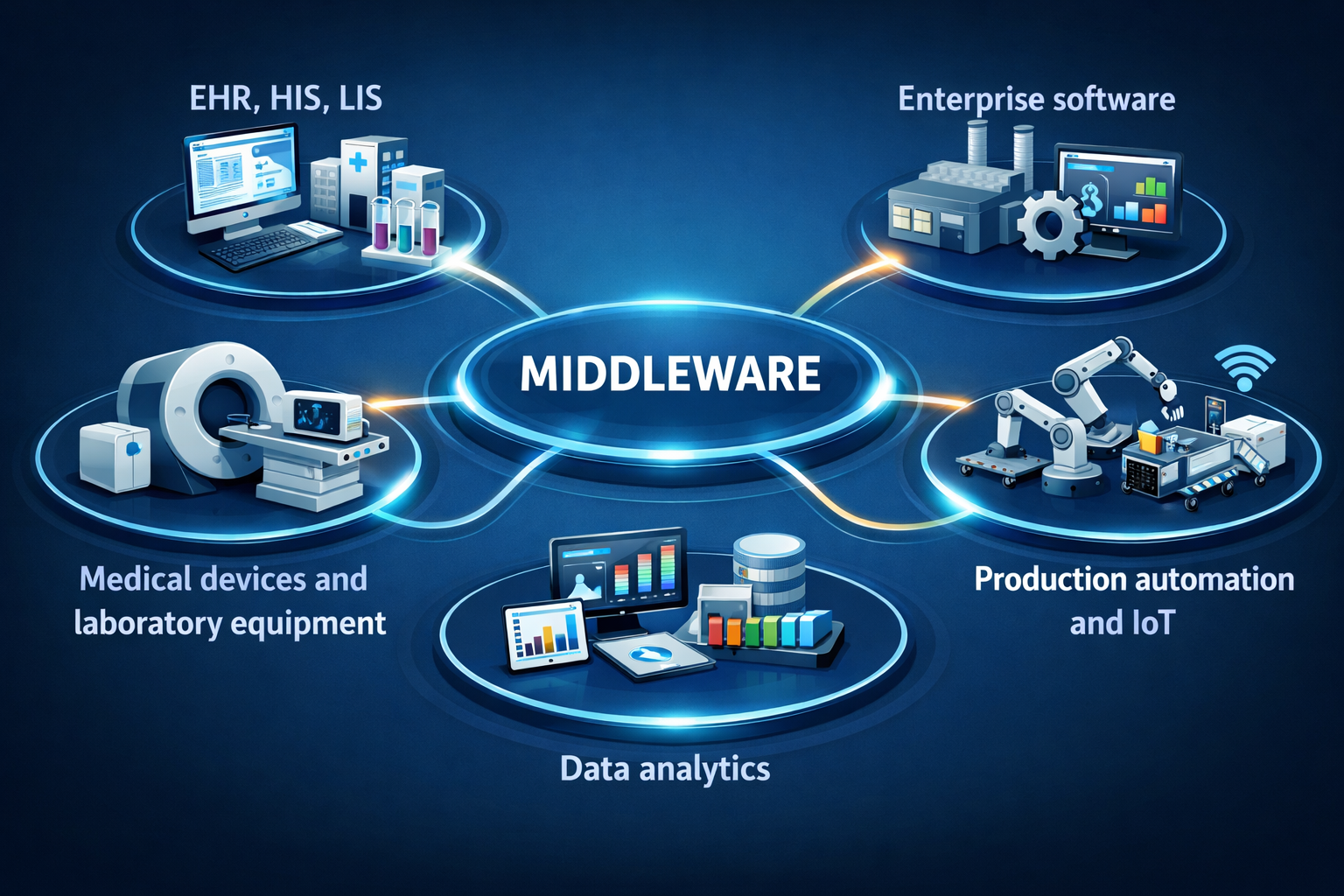
Implementing middleware is not a mere technical deployment; it is an architectural transformation. Middleware becomes the backbone connecting:
- Clinical systems (EHR, HIS, LIS)
- Enterprise software (ERP, CRM, MES)
- Medical devices and laboratory equipment
- Production automation and IoT systems
- Data analytics platforms
By orchestrating these systems, middleware allows organizations to move from fragmented workflows to integrated, proactive operations, enhancing clinical decision-making, operational efficiency, and regulatory compliance.
Organizations evaluating modernization initiatives often begin by exploring enterprise-grade medical integration middleware. A dedicated platform provides visibility into system interoperability, reduces operational complexity, and ensures scalability for future healthcare innovations
Understanding the Scope of Middleware Implementation
Middleware implementations are rarely simple point-to-point connections. They typically orchestrate communication between multiple systems, devices, and protocols across healthcare and production environments.
Common Integration Targets
- Enterprise Systems: EHR, HIS, ERP, MES, LIS
- Medical Devices & Equipment: Diagnostic machines, imaging devices, analyzers, pharmacy automation, robotic systems
- Communication Protocols: HL7, FHIR, REST APIs, SOAP, OPC UA, Modbus, DICOM
Middleware manages these environments through:
- Protocol translation
- Data routing
- Message validation
- Event orchestration
- Error handling
The result is a centralized, reliable integration layer that allows legacy and modern systems to function as a coordinated ecosystem.
Phase 1 — Discovery and Integration Strategy
The discovery phase lays the foundation for every successful middleware implementation. During this stage, organizations gain visibility into existing systems, workflows, and integration requirements.
Without comprehensive discovery, integration projects risk:
- Unexpected system dependencies
- Data inconsistencies
- Workflow disruptions
- Performance bottlenecks
System Landscape Analysis
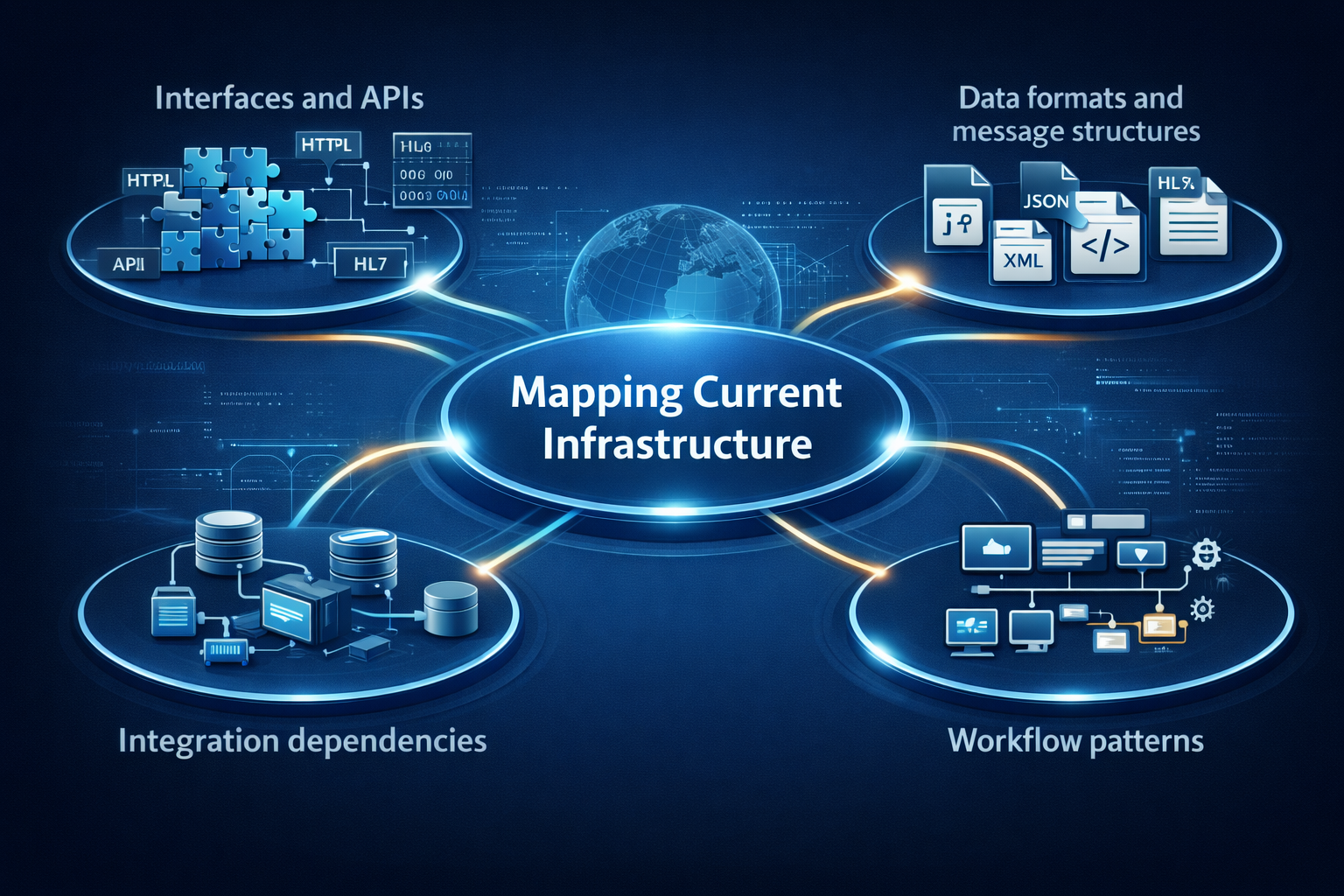
Discovery begins with mapping the current infrastructure:
- Interfaces and APIs
- Data formats and message structures
- Integration dependencies
- Workflow patterns
This analysis identifies legacy constraints, inefficiencies, and potential points of failure.
Device and Hardware Assessment
Healthcare and production environments rely on specialized devices. During discovery, teams evaluate:
- Communication protocols and firmware capabilities
- Data transmission methods
- Driver compatibility
- Telemetry and connectivity limitations
This ensures integration strategies account for device behavior and real-world operational conditions.
Workflow Mapping
Workflow mapping identifies:
- How data flows between systems
- Manual bottlenecks
- Automation opportunities
Understanding workflows allows middleware to orchestrate operations without disrupting existing clinical or production processes.
Risk Identification
Common risks discovered during discovery include:
- Legacy system limitations
- Protocol mismatches
- Data inconsistencies
- Network and infrastructure constraints
Addressing these risks upfront is critical for project success.
Implementation timelines vary based on complexity, number of systems, and device diversity. While small-scale integrations may be completed within weeks, large enterprise environments with multiple systems and regulatory constraints may require several months. Structured discovery helps produce realistic timelines and avoid delays.
Many organizations benefit from early technical assessments that identify gaps, risks, and modernization opportunities. Evaluating middleware readiness at the discovery stage reduces risk and accelerates implementation.
Phase 2 — Architecture Design and System Blueprinting
After discovery, architects design the middleware architecture, translating requirements into technical blueprints. Architecture design defines how systems communicate, how data flows, and how reliability is maintained.
Middleware Topology
Middleware topology determines system organization:
- Centralized hubs
- Distributed nodes
- Hybrid cloud and on-premises architectures
Topology decisions are based on system complexity, data volume, latency requirements, and compliance obligations.
Most legacy systems do not require replacement. Middleware enables connectivity through adapters, protocol translation, and transformation layers, allowing incremental modernization without disrupting operations.
Data Routing and Transformation
Middleware translates messages between protocols, ensuring interoperability between systems that use HL7, FHIR, REST APIs, and other standards. Data routing strategies maintain accuracy, consistency, and operational efficiency.
Fault Tolerance and Reliability
High system availability is crucial. Middleware architectures incorporate:
- Message queuing
- Failover and redundancy
- Retry logic
- Audit logging
These measures minimize downtime and prevent data loss.
Security and Compliance

Enterprise middleware designs include:
- Secure authentication
- End-to-end encryption
- Role-based access controls
- Audit trails for compliance
These features ensure that the integration layer meets stringent healthcare regulations.
Engaging experienced integration architects at this stage ensures infrastructure decisions align with operational and regulatory requirements, reducing costly redesigns later.
Phase 3 — Development of Integration Components
Development focuses on building the integration components that connect systems and devices.
Custom Adapters
Adapters act as bridges between middleware and specific systems, including:
- Legacy platforms
- Medical devices
- Enterprise software
- External services
Custom adapters handle unique communication protocols and workflows.
Protocol Translation
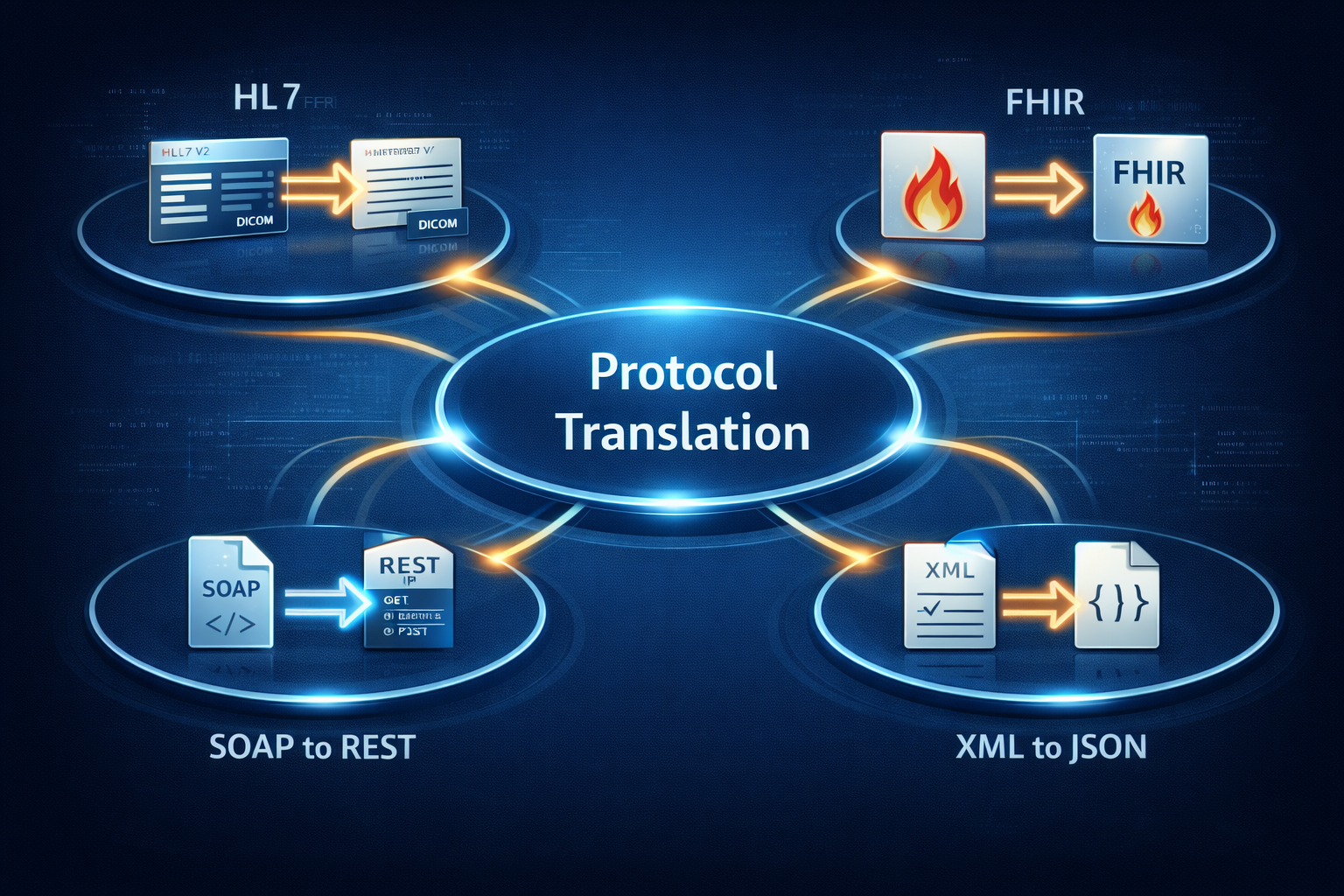 Middleware converts messages between formats to enable interoperability. Common transformations include:
Middleware converts messages between formats to enable interoperability. Common transformations include:
- HL7 to FHIR
- SOAP to REST
- XML to JSON
Enterprise middleware platforms are designed to handle multiple protocols, allowing heterogeneous systems to coexist in a unified integration environment.
Data Validation and Normalization
Validation rules ensure data completeness, correctness, and consistency. Normalization aligns data across systems to prevent discrepancies.
Logging and Monitoring
Robust logging and monitoring enable teams to track message delivery, error rates, and performance metrics, improving troubleshooting and system observability.
Organizations prioritizing adapter development, validation, and observability frameworks achieve faster adoption and more predictable integration outcomes.
Phase 4 — Testing, Validation, and Compliance
Testing ensures middleware operates reliably in real-world conditions.
Integration Testing
Verifies correct communication and workflow execution across systems.
Performance Testing
Assesses throughput, latency, and scalability under expected operational loads.
Failover and Recovery Testing
Simulates network or system failures to ensure continuous operation.
Compliance Validation
Ensures data integrity, auditability, and regulatory adherence.
Integrated FAQ:
How is data accuracy maintained during integration?
Data is validated at multiple layers, with automated testing simulating real workflows to detect discrepancies before production. This minimizes errors and ensures regulatory compliance.
CTA:
Structured validation strategies reduce operational risks and improve confidence during go-live transitions.
Phase 5 — Deployment and Go-Live
Deployment transitions middleware from testing to production.
Controlled Rollout
Enterprises often use pilot deployments, phased rollouts, or parallel operations to minimize disruption.
Parallel Validation
Running legacy and new systems together confirms data consistency and workflow reliability before fully transitioning.
Real-Time Monitoring
Monitoring during go-live ensures immediate visibility into system health, message processing, and errors.
Expect active monitoring, staged deployment, and close coordination between IT, operations, and clinical teams to ensure seamless integration.
Structured rollout strategies with real-time monitoring increase operational continuity and minimize disruptions during deployment.
Phase 6 — Post-Go-Live Stabilization and Optimization
Even after deployment, middleware requires ongoing attention.
Performance Tuning
Adjust routing, resources, and transformation logic to optimize efficiency.
Continuous Monitoring
Track system health, errors, and message flow to maintain stability.
Scaling
Support growth by adding processing nodes, expanding cloud resources, and upgrading infrastructure.
Middleware is an evolving capability. Planning for scalability ensures the environment remains adaptable and future-ready.
Measuring Success
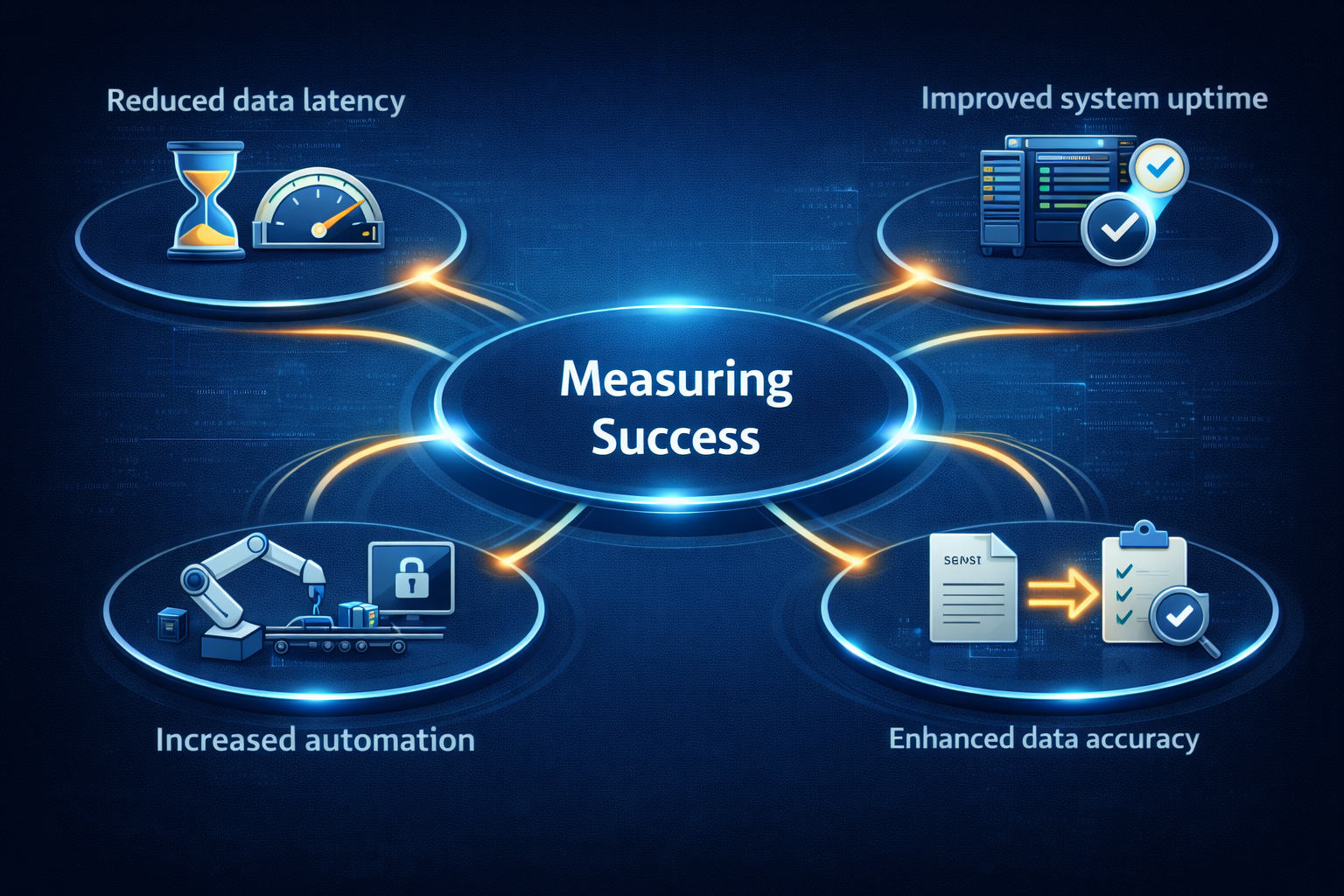
Key performance indicators include:
- Reduced data latency
- Improved system uptime
- Increased automation
- Fewer manual interventions
- Enhanced data accuracy
These metrics demonstrate ROI and validate modernization efforts.
Long-Term Healthcare Modernization
Middleware implementation is a foundation for future capabilities:
- AI-driven decision support
- Predictive analytics
- Remote monitoring
- Intelligent workflow automation
Organizations that invest in scalable integration architecture today are positioned to adopt emerging technologies without disruptive overhauls.
Conclusion
Medical integration middleware is a strategic enterprise asset, not just a technical solution. From discovery through post-go-live optimization, a structured lifecycle ensures risk mitigation, operational continuity, and modernization readiness.
Enterprises that prioritize architecture-first design, rigorous testing, and ongoing optimization position themselves for long-term success in connected healthcare ecosystems.
To explore how enterprise-grade medical integration middleware supports scalable, compliant, and future-ready healthcare infrastructure, review the full solution overview.
Categories
Share
Need a project estimate?
Drop us a line, and we provide you with a qualified consultation.




